Peritoneal Anatomy
The peritoneum is a thin,
translucent,
serous membrane.
The parietal and visceral peritoneum are the two layers of peritoneum.
The peritoneal cavity is a potential space between the two layers.
Peritoneal ligaments are double layers or folds of peritoneum that support a structure within the peritoneal cavity; omentum and mesentery are specifically named peritoneal ligaments.
Omentum is a double layer of peritoneum that extends between the stomach and duodenal bulb to adjacent organs.
Mesentery is a double layer of peritoneum that encloses an organ and connects it to the abdominal wall.
The mesenteric contains the blood vessels,
lymph nodes,
nerves,
and fat.
The mesentery divides the peritoneal cavity into various spaces.
The pathways of disease spread in the abdominal cavity
Subperitoneal – All the organs are subperitoneal thus it is an anatomical pathway through which there is spread of tumour.
The subperitoneal space is continuous between organ,
thus there is spread of tumour from one to another
Potential routes - along the mesenteries and ligaments,
via visceral lymphatics to nodes,
& by periarterial,
perineural,
or transvenous routes as well as along ducts.
Transperitoneal – Spread occurs when disease traverses the visceral peritoneum.
Neoplastic and inflammatory cells,
gas,
and haemorrhage in the subperitoneal space can therefore,
cross the peritoneum to enter the peritoneal cavity.
Subsequently,
peritoneal spread occurs via the circulating peritoneal fluid to the peritoneal recesses in the abdomen and pelvis.
Flow of peritoneal fluid
The low subdiaphragmatic pressure draws the peritoneal fluid from the pelvis into the abdomen.
Fluid (and cancer cells) travels from the pelvis to the abdomen via the paracolic gutters and travels from the abdominal infracolic compartment to the pelvis
Fluid pools in dependent recesses like the pouch of Douglas,
along the superior portion of the sigmoid mesocolon,
ileocolic region,
right paracolic gutter,
and Morison’s pouch
Although the falciform and phrenicocolic ligaments typically limit fluid flow across them,
large volumes of fluid can overflow under the free edge of the falciform ligament and over the phrenicocolic ligament.
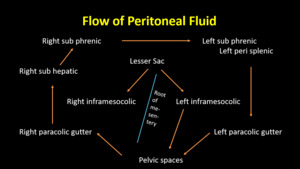
Fig. 1
Peritoneal spaces in relation to Peritoneal carcinomatosis index
The abdomen and pelvis is divided into 13 regions (numbered 0-12).
Two transverse and two sagittal planes divide the abdomen into 9 regions,
numbered in a clockwise direction - 0 at the umbilicus and 1 defining the space beneath the right hemidiaphragm
Regions 9 through 12 divide the small bowel into upper and lower jejunum and upper and lower ileum
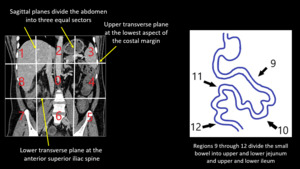
Fig. 2: Peritoneal regions according to PCI.
References: Rhonda L Harmon and Paul H Sugarbaker, Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer, Int Semin Surg Oncol. 2005; 2: 3.
Peritoneal carcinomatosis index is a quantitative indicator of peritoneal disease.
Quantitative prognostic indicators are to serve as guidelines in the selection of treatments to maximize the benefits of therapy and to exclude patients who have little or no chance to improve.
The various indicators that are used are -
1.Intraoperative assessment of the extent of carcinomatosis at time of surgical exploration
• Gilly peritoneal carcinomatosis staging
• Carcinomatosis staging by the Japanese Research Society for Gastric Cancer
• Peritoneal Cancer Index (PCI)
• Dutch Simplified Peritoneal Carcinomatosis Index (SPCI)
2.CT PCI
3.Prior Surgical Score
4.Completeness of Cytoreduction Score
Intraoperative assessment of peritoneal disease.
Gilly Peritoneal Carcinomatosis Staging index and Carcinomatosis staging by the Japanese Research Society for Gastric Cancer were used initially and had their limitations.
Peritoneal Carcinomatosis index has replaced them all and is used universally.
Peritoneal Carcinomatosis Index
The Peritoneal Carcinomatosis Index (PCI) is determined at the time of surgical exploration of the abdomen and pelvis.
It is a quantitative assessment of the distribution of tumour in the abdominal cavity.
PCI is a prognostic indicator and also helps in assessing the volume of tumour burden and the likelihood of complete CRS.
The abdominal cavity is divided into 13 regions as described above.
Each region is given a number according to the lesion size of the deposit.
The lesion size (LS) score refers to the greatest diameter of tumour implants that are distributed on the peritoneal surfaces.
Primary tumours or localized recurrences at the primary site that can be removed definitively are excluded from the assessment.
The lesion sizes are then summated for all abdominopelvic regions.
The extent of the disease within all regions of the abdomen and pelvis is indicated by a numerical score from 0 to 39.

Table 1: Peritoneal anatomical regions according to PCI and the lesion size score
References: Rhonda L Harmon and Paul H Sugarbaker, Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer, Int Semin Surg Oncol. 2005; 2: 3.
Dutch Simplified Peritoneal Carcinomatosis Index (SPCI)- It is similar to PCI.
It also has a correlate to the index and the rate of complications for combined treatment.
However,
PCI is more commonly used.
Role of Imaging in the assessment of PCI
Diagnostic laparoscopy is the gold standard for PCI.
The preoperative CT is an excellent tool in locating and quantifying mucinous adenocarcinoma within the peritoneal cavity.
However,
it is less sensitive in picking up lesions less than 5mm in size.
Combining PET CT with CT increases the accuracy of determining PCI.
CE MRI provides better soft tissue contrast,
multiphase dynamic contrast images and DWI.
MRI shows increased conspicuity of small lesions and peritoneal disease shows delayed enhancement thus more conspicuous on delayed post-contrast MR scans.
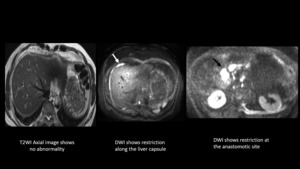
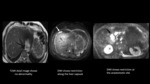
Fig. 3: History of periampullary carcinoma, post treatment, on surveillance with raised tumor markers. Patient had a normal CT (not shown). T2WI reveals no abnormality. However restriction seen on DWI suggestive of recurrent perihepatic and anastomotic site disease.
References: Christian Medical College, Vellore, Tamil Nadu, India
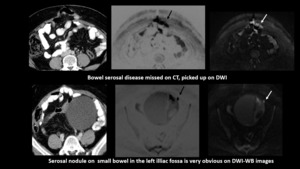
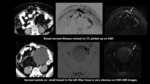
Fig. 4: In a case of carcinoma ovary,post operative & post chemotherapy with residual disease.She was being considered for
HIPEC. Axial CT scan missed the disease. However, DWI and their inverted images picked up the lesions showing restricted diffusion
References: Christian Medical College, Vellore, Tamil Nadu, India
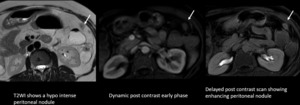
Fig. 5: Case of carcinoma sigmoid colon MRI revealed a PCI score of 12.
The peritoneal lesion is better appreciated on delayed post contrast TIW fat suppressed image
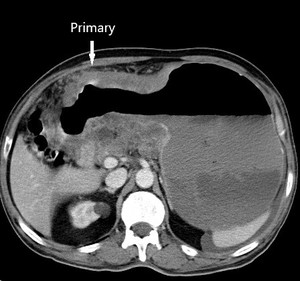
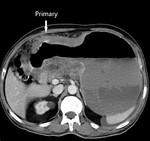
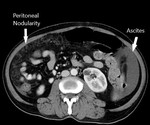
Fig. 6: Case of gastric carcinoma with serosal involvement and peritoneal disease and ascites.Axial CECT scan shows irregular thickening of the stomach antrum and lesser curvature.
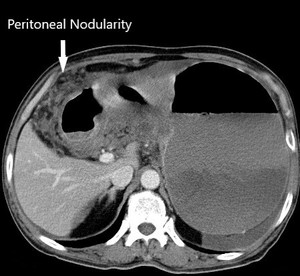
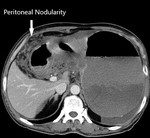
Fig. 7: Peritoneal nodularity seen in the same patient.
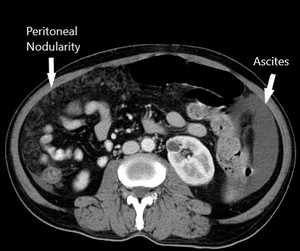
Fig. 8: Peritoneal nodularity and ascites in the same patient. The CT PCI is >7, which is a contra-indication to HIPEC in our institute. The patient was started on palliative chemotherapy.
Steps for peritoneal disease assessment -
The following are the steps of peritoneal disease assessment-

Fig. 9: Steps for peritoneal disease assessment
References: Emily A. Aherne et al, What the Radiologist Should Know About Treatment of Peritoneal Malignancy, American Journal of Radiology, March 2017, Volume 208, Number 3
PAUSE
PAUSE is used for describing key findings in peritoneal disease in a radiology report.
Its an acronym for -
P – Primary tumour and peritoneal carcinomatosis index (PCI)
A – Ascites and abdominal wall involvement
U – Unfavourable sites of involvement(eg.
Common bile duct)
S – Small bowel and mesenteric disease
E – Extra peritoneal metastases
Calculating PCI in a case of carcinoma ascending colon -
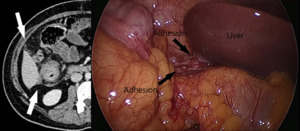
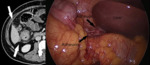
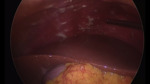
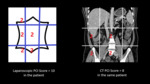
Fig. 10: Laparoscopic image of the right sub-hepatic region involvement with adhesion and corresponding CT in a case of Carcinoma of Ascending Colon.
Score - 2
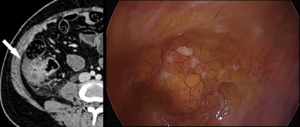
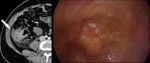
Fig. 11: Laparoscopic image of the right flank region and corresponding CT of the same patient. Score-2
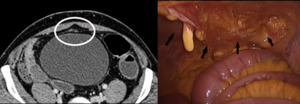
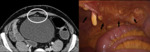
Fig. 12: Laparoscopic image of the right umbilical and hypogastric region and corresponding CT of the same patient. Score-2+2
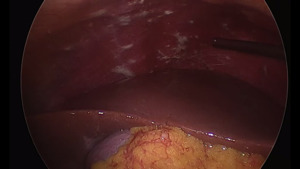
Fig. 13: Sub diaphragmatic involvement was also seen on Laparoscopy; not appreciated on CT
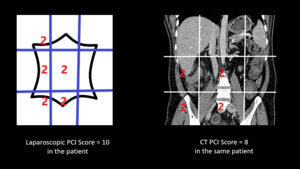
Fig. 14: Thus the total PCI scores of the above patient are : on Laproscopy - 10 and on CT - 8
Role of PCI in decision making
Management of peritoneal disease is a combination of surgery and intraperitoneal chemotherapy.
Different institutes use different cut-offs of PCI for using combined therapy.
The cut-offs vary depending on the primary cancer and its histology.
Involvement of a vital structure such as the root of mesentery,
liver hilum renders the patient inoperable and must be reported.
The extent of small bowel involvement is also important to prevent short gut syndrome.
The PCI cut-offs used at our institute are as follows -
- Colorectal carcinoma: 10-15; depending on patient factors
- Carcinoma stomach: 5-9; depending on patient factors
- Ovarian tumours,
pseudomyxoma peritonei and minimally aggressive mesothelioma: No cutoff as long as cytoreduction possible.
The aim of CRS is to eliminate all macroscopic disease through a series of visceral resections followed by targeting any residual microscopic disease with intraperitoneal chemotherapy,
exposing the peritoneal surfaces to a high concentration of chemotherapy with lower systemic toxicity.
CRS – Cytoreductive surgery comprises of peritonectomy procedures and visceral resections.
The goal is to remove all macroscopic disease leaving no residual disease.
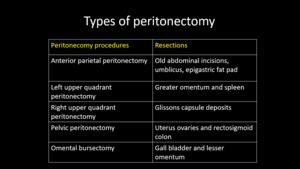
Table 2: Types of peritonectomy procedures
References: . Sanket S. Mehta et al, Cytoreductive Surgery and Peritonectomy Procedures, Indian J Surg Oncol. 2016 Jun; 7(2): 139–151.
There is no standardized methodology for intraperitoneal chemotherapy administration.
Different regimens of intraperitoneal chemotherapy include –
1 HIPEC
2 EPIC
4 Bidirectional chemotherapy
5 NIPS
6 PIPAC
Among these HIPEC is the most commonly used.
1.HIPEC– Hyperthermic intraperitoneal chemotherapy is delivered after the surgical procedure in the operating room.
It is delivered once CRS has been completed before any digestive reconstruction or diversion is made.
Moderate hyperthermia (above 41°C) increases penetration and cytotoxicity of chemotherapeutic agent.
CRS without HIPEC leads to entrapment of microscopic disease with early recurrence.
HIPEC after post-surgical recovery leads to non-uniform distribution of drug due to adhesions.
Mitomycin C is the HIPEC drug of choice in patients with colorectal and appendiceal primary,
and in a subset of patients with mesothelioma.
Cisplatin HIPEC has been used in mesothelioma,
ovarian and gastric cancer.
However,
it is associated with increased toxicity and complications.
Mode of delivering HIPEC is by closed and open methods.
- In open method,
the abdomen is kept open using retractors and chemotherapeutic agent is poured into the abdominal cavity.
- In closed method,
the abdomen is closed with sutures and two drains inserted for inflow and outflow of chemotherapeutic agent.
The open method is the best technique for HIPEC delivery as per the consensus statement issued by the Peritoneal Surface Oncology Group International,
2006
2.EPIC– Early post- operative chemotherapy is given via a port in a post-surgical setting on day 1-5.
It does not involve hyperthermia and can be administered in multiple settings.
There is an increased risk of systemic absorbtion and adverse effects,
as the chemotherapeutic agents remain in the peritoneal cavity.
3.Bidirectional chemotherapy– It combines both intraperitoneal chemotherapy and systemic chemotherapy targeting both the peritoneal cavity and the subperitoneal blood vessels.
Intravenous drugs can be targeted to the peritoneal surface if administered simultaneously with a large volume of intraperitoneal chemotherapy solution.
It is used in colorectal and appendix adenocarcinoma.
4.NIPS-Neoadjuvant Intraperitoneal and Systemic Chemotherapy is an intravenous and intraperitoneal chemotherapy regimen given as an option for reducing the tumour load prior to CRS and therefore may even facilitate definitive CRS
Post NIPS increased fibrosis,
thus causing difficult surgery.
Useful in the management of peritoneal metastases from gastric cancer.
5.PIPAC- Pressurized intraperitoneal aerosol chemotherapy is a novel technique delivering drugs into the abdominal cavity as an aerosol under pressure.
It generates an artificial pressure gradient,
enhancing tissue uptake and better homogenous distribution.
PIPAC is a short laparoscopic procedure than HIPEC and can be repeated.
It is a palliative intent procedure,
no resection is performed; hence morbidity and mortality is low,
and the hospital stay is short
It is done for patients whose PCI is high for HIPEC and disease is unresectable.